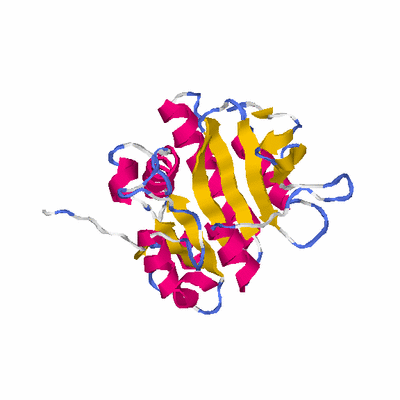
##########
# PROTEIN: Q2246_545_124.5wLII_11077_30 266 124 # Category_B
# Length: 266
# Weight: 30217.92
# Similarity: UniProt_B6ARD8, BLAST_B6ARD8, PDB_templates_INFO
# Best Pcover Model PDB N_AA SISC E-val Seq_ID LAL Overlap
# Cat 78.95 M_26 2qvp_C 275 3 8e-36 25.000 210:238 (36-261:50-271)
# Evl 81.95 M_00 2boa_A 404 2 1e-58 15.000 218:264 (4-225:106-365)
# Sid 24.81 M_33 2qvp_C 275 3 8e-06 37.000 66:70 (53-122:66-131)
# Cov 98.12 M_03 1h8l_A 380 2 1e-48 12.000 261:296 (3-264:9-303)
# LaL 28.57 M_36 1cpb_A 82 1 2e-20 18.000 76:77 (2-78:7-82)
# OvN 94.74 M_00 1kwm_A 402 2 1e-57 14.000 252:307 (2-260:102-401)
# OvC 89.85 M_12 1h8l_A 380 2 2e-52 13.000 239:298 (3-266:9-281)
PDB: 2qvp PDBsum
HEADER STRUCTURAL GENOMICS, UNKNOWN FUNCTION 08-AUG-07 2QVP
TITLE CRYSTAL STRUCTURE OF UNCHARACTERIZED PROTEIN (YP_926603.1)
TITLE 2 FROM SHEWANELLA AMAZONENSIS SB2B AT 2.00 A RESOLUTION
SOURCE 2 ORGANISM_SCIENTIFIC: SHEWANELLA AMAZONENSIS SB2B;
SOURCE 3 ORGANISM_TAXID: 326297;
KEYWDS YP_926603.1, UNCHARACTERIZED PROTEIN, STRUCTURAL GENOMICS,
KEYWDS 2 JOINT CENTER FOR STRUCTURAL GENOMICS, JCSG, PROTEIN
KEYWDS 3 STRUCTURE INITIATIVE, PSI-2, HYDROLASE, UNKNOWN FUNCTION
SCOP: none
PDB: 2boa PDBsum
HEADER HYDROLASE 08-APR-05 2BOA
TITLE HUMAN PROCARBOXYPEPTIDASE A4.
SOURCE 2 ORGANISM_SCIENTIFIC: HOMO SAPIENS;
SOURCE 3 ORGANISM_COMMON: HUMAN;
SOURCE 4 ORGANISM_TAXID: 9606;
KEYWDS METALLOPROCARBOXYPEPTIDASE, X-RAY CRYSTAL STRUCTURE,
KEYWDS 2 ZYMOGEN, METALLOPROTEASE, EXOPROPEPTIDASE, HYDROLASE,
KEYWDS 3 CARBOXYPEPTIDASE
SCOP: c.56 Phosphorylase/hydrolase-like
SCOP: c.56.5 Zn-dependent exopeptidases
SCOP: c.56.5.1 Pancreatic carboxypeptidases
SCOP: d.58 Ferredoxin-like
SCOP: d.58.3 Protease propeptides/inhibitors
SCOP: d.58.3.1 Pancreatic carboxypeptidase, activation domain
PDB: 1h8l PDBsum
HEADER CARBOXYPEPTIDASE 09-FEB-01 1H8L
TITLE DUCK CARBOXYPEPTIDASE D DOMAIN II IN COMPLEX WITH GEMSA
SOURCE 2 ORGANISM_SCIENTIFIC: LOPHONETTA SPECULARIOIDES;
SOURCE 3 ORGANISM_COMMON: CRESTED DUCK;
SOURCE 4 ORGANISM_TAXID: 8836;
KEYWDS CARBOXYPEPTIDASE, HYDROLASE, ZINC-DEPENDENT PROTEASE
SCOP: b.3 Prealbumin-like
SCOP: b.3.2 Carboxypeptidase regulatory domain-like
SCOP: b.3.2.1 Carboxypeptidase regulatory domain
SCOP: c.56 Phosphorylase/hydrolase-like
SCOP: c.56.5 Zn-dependent exopeptidases
SCOP: c.56.5.1 Pancreatic carboxypeptidases
PDB: 1cpb PDBsum
HEADER HYDROLASE (C-TERMINAL PEPTIDASE) 23-JUN-76 1CPB
TITLE STRUCTURE OF CARBOXYPEPTIDASE B AT 2.8 ANGSTROMS RESOLUTION
SOURCE 2 ORGANISM_SCIENTIFIC: BOS TAURUS;
SOURCE 3 ORGANISM_COMMON: CATTLE;
SOURCE 4 ORGANISM_TAXID: 9913;
COMPND 4 EC: 3.4.17.2;
COMPND 9 EC: 3.4.17.2;
KEYWDS HYDROLASE (C-TERMINAL PEPTIDASE)
SCOP: c.56 Phosphorylase/hydrolase-like
SCOP: c.56.5 Zn-dependent exopeptidases
SCOP: c.56.5.1 Pancreatic carboxypeptidases
PDB: 1kwm PDBsum
HEADER HYDROLASE 30-JAN-02 1KWM
TITLE HUMAN PROCARBOXYPEPTIDASE B: THREE-DIMENSIONAL STRUCTURE
TITLE 2 AND IMPLICATIONS FOR THROMBIN-ACTIVATABLE FIBRINOLYSIS
TITLE 3 INHIBITOR (TAFI)
SOURCE 2 ORGANISM_SCIENTIFIC: HOMO SAPIENS;
SOURCE 3 ORGANISM_COMMON: HUMAN;
SOURCE 4 ORGANISM_TAXID: 9606;
COMPND 4 EC: 3.4.17.2;
KEYWDS HYDROLASE, PROCARBOXYPEPTIDASE B
SCOP: c.56 Phosphorylase/hydrolase-like
SCOP: c.56.5 Zn-dependent exopeptidases
SCOP: c.56.5.1 Pancreatic carboxypeptidases
SCOP: d.58 Ferredoxin-like
SCOP: d.58.3 Protease propeptides/inhibitors
SCOP: d.58.3.1 Pancreatic carboxypeptidase, activation domain